Abstract
Introduction: Diffuse large B-cell lymphoma (DLBCL) represents 33% of the non-Hodgkin lymphomas (NHL) and expresses CD19, a classic B-cell marker found on B lymphocytes. ADCT-402 (loncastuximab tesirine; Lonca-T) is an antibody drug conjugate comprising a humanized antibody directed against human CD19 conjugated to a pyrrolobenzodiazepine dimer toxin. This first-in-human clinical study evaluated the safety and efficacy of Lonca-T in patients (pts) with relapsed/refractory (R/R) B-cell lineage NHL. Here we present interim results in the subgroup of pts with DLBCL. Interim efficacy and safety of Lonca-T in pts with follicular lymphoma and mantle cell lymphoma are presented in a separate abstract.
Methods: Pts ≥18 years of age with R/R DLBCL who have failed or are intolerant to established therapies, or have no other treatment options available, were enrolled in this Phase 1, multicenter, open-label, single-arm study, including dose-escalation and dose-expansion parts. The primary objectives are to evaluate the safety and tolerability of Lonca-T, and determine the recommended dose(s) to use for expansion cohorts. The secondary objectives are to evaluate the clinical activity (measured by overall response rate [ORR], duration of response [DoR], progression-free survival [PFS] and overall survival [OS]), pharmacokinetics, pharmacodynamics, and anti-drug antibody activity. Pts receive 1-hour intravenous infusions of Lonca-T every 3 weeks (1 cycle), with a 3+3 dose-escalation design for the dose-escalation part of the study. No intra-pt dose escalation is allowed.
Results: As of June 20, 2018, 183 pts had been enrolled on the study, including 137 with DLBCL (79 male, 58 female). Pts with DLBCL had a median age of 63 years [range 20-86], and had received a median of
3 previous therapies (range 1-10; Table). Pts received doses of Lonca-T ranging from 15 to 200 µg/kg (median cycles: 2 [range 1-13]). Treatment-emergent adverse events (TEAEs) were reported in 136/137 (99.3%) pts, and grade ≥3 TEAEs in 100/137 (73.0%) pts. The most common all-grade TEAEs (≥20% pts), regardless of relationship to study treatment, were fatigue (57 [41.6%]), nausea (44 [32.1%], peripheral edema (44 [32.1%]), anemia (39 [28.5%]), rash (35 [25.5%]), gamma-glutamyltransferase (GGT) increased (33 [24.1%]), constipation (30 [21.9%]), dyspnea (29 [21.2%]), and thrombocytopenia (28 [20.4%]). The most common grade ≥3 TEAEs (>10% pts) were GGT increased (21 [15.3%]), neutropenia (20 [14.6%]), neutrophil count decreased (19 [13.9%]), anemia (15 [10.9]), thrombocytopenia (15 [10.9%]) and platelet count decreased (14 [10.2%]. Approximately 66% and 72% of pts in the 120 and 150 µg/kg groups, respectively, tolerated at least 2 cycles before any AE leading to dose reduction/delay occurred.
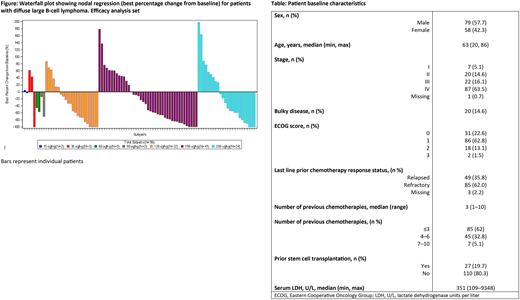
The figure depicts tumor response data. Out of 132 evaluable pts with DLBCL, the ORR was 40.2% (53/132 pts), comprising 29/132 (22.0%) complete responses (CRs) and 24/132 (18.2%) partial responses (PRs). Median DoR was 4.17 months and PFS was 2.79 months after a median follow-up of 5.13 months. Median DoR was not reached in pts achieving a CR and was 2.76 months in pts with a PR. In pts with non-bulky disease, the ORR was 44.2% (50/113 pts); 28/113 (24.8%) pts attained a CR and 22/113 (19.5%) pts attained a PR. The majority of pts (122/132) received doses ≥120 µg/kg; in these pts, the ORR was 41.8% (51/122 pts), with 28/122 (23.0%) pts attaining a CR and 23/122 (18.9%) pts attaining
a PR.
Conclusions: In this Phase 1 study, Lonca-T has demonstrated encouraging and durable single-agent antitumor activity and manageable toxicity in pts with R/R DLBCL at doses ≥120 µg/kg. Updated safety, tolerability, and efficacy results will be presented at the meeting.
Study sponsored by ADC Therapeutics. http://clinicaltrials.gov/show/NCT02669017.
Radford:Pfizer: Research Funding; ADC Therapeutics: Consultancy, Research Funding; BMS: Consultancy, Speakers Bureau; GlaxoSmithKline: Equity Ownership; Seattle Genetics: Consultancy, Speakers Bureau; Takeda: Consultancy, Research Funding, Speakers Bureau; AstraZeneca: Equity Ownership; Novartis: Consultancy, Speakers Bureau; Celgene: Research Funding. Kahl:Seattle Genetics: Consultancy; Genentech: Consultancy; ADC Therapeutics: Research Funding. Hamadani:Sanofi Genzyme: Research Funding, Speakers Bureau; Takeda: Research Funding; Cellerant: Consultancy; Ostuka: Research Funding; MedImmune: Consultancy, Research Funding; ADC Therapeutics: Research Funding; Janssen: Consultancy; Celgene Corporation: Consultancy; Merck: Research Funding. Carlo-Stella:Boehringher Ingelheim Italia: Consultancy; Bristol-Myers Squibb: Speakers Bureau; Sanofi: Consultancy; MSD Italia: Speakers Bureau; Rhizen Pharmaceuticals: Research Funding; Genenta Science: Speakers Bureau; Janssen: Speakers Bureau; AstraZeneca: Speakers Bureau; Amgen: Speakers Bureau; ADC Therapeutics: Research Funding, Speakers Bureau. Caimi:Genentech: Membership on an entity's Board of Directors or advisory committees; ADC Therapeutics: Research Funding; Celgene: Speakers Bureau; Kite Pharma: Membership on an entity's Board of Directors or advisory committees. Ardeshna:Roche: Membership on an entity's Board of Directors or advisory committees, Other: Conference expenses, Research Funding; Celgene: Membership on an entity's Board of Directors or advisory committees; ADC Therapeutics: Membership on an entity's Board of Directors or advisory committees, Research Funding; Takeda: Membership on an entity's Board of Directors or advisory committees. Feingold:ADC Therapeutics: Employment, Equity Ownership. He:ADC Therapeutics: Employment, Equity Ownership. Reid:AbbVie: Research Funding; Millenium Pharmaceuticals: Research Funding; ADC Therapeutics: Research Funding. Solh:ADC Therapeutics: Research Funding; Celgene: Speakers Bureau; Amgen: Speakers Bureau. Chung:ADC Therapeutics: Research Funding. Heffner:Pharmacyclics: Research Funding; ADC Therapeutics: Research Funding; Kite Pharma: Research Funding; Genentech: Research Funding. Ungar:ADC Therapeutics: Employment, Equity Ownership. O'Connor:ADC Therapeutics: Research Funding; Seattle Genetics: Research Funding; Celgene: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal